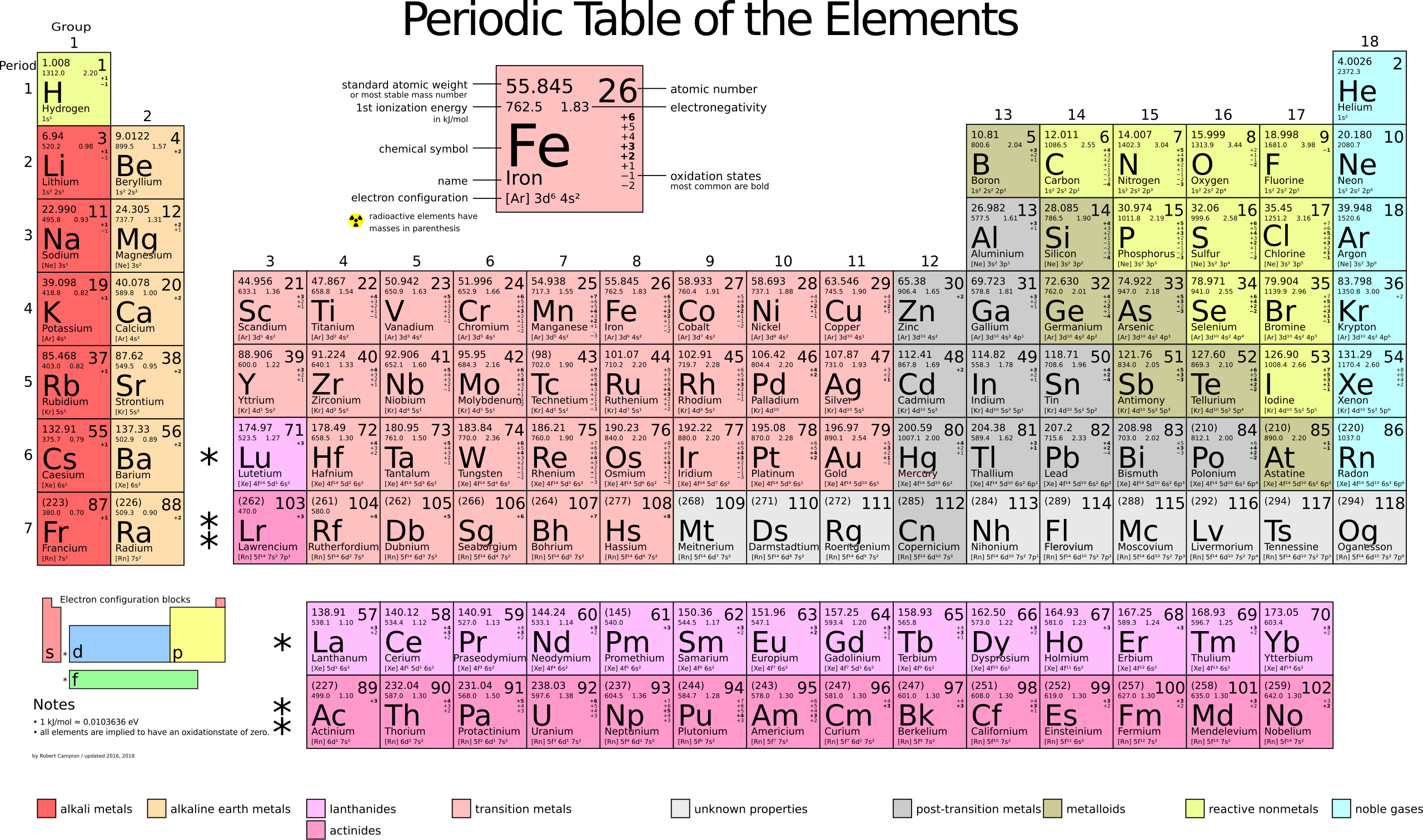
What to Use for Memorization
Use the Major System to link the atomic number with the name of the element (1 with Hydrogen, 2 with Helium, 3 with Lithium, etc.).
Examples:
- Linking 1 with Hydrogen (“1” with “H”): Use the word associated with the number 1 in the Major System. If it’s “Tea”, then create a phrase with “Hydrogen”: “Tea burns with Hydrogen”.
- Linking 13 with Aluminum: Use “time” (13 in the Major System) and “Aluminum”: “Gave the time with aluminium foil”.
Deepening Your Memorization of the Periodic Table
To enhance the way you memorize the elements of the Periodic Table, add more information like:
- Chemical Symbol: H for Hydrogen, He for Helium, Na for Sodium, Li for Lithium
- Atomic Weight
- Original Name: Gold (Aurum), Antimony (Stibium), etc.
- Melting Point
- Boiling Point
- Density
- Acid/Base Properties
Ideas to Increase Data Memorization Using the Link Method:
- Chemical Symbol (H): The “Tea” (1) could take the shape of a giant “H”.
- Atomic Weight (1.00794 g/mol): Link the “Tea” or bag of “Tea” or “auto” or “fairy” (for “1”) burning Hydrogen, eating a “River” and another “River” (for “00”) of news from the “EFE” news agency (for “7”) with its enormous “mouth” (for “94”).
Other Methods for Memorizing the Periodic Table:
- Using Mind Maps: Create a Mind Map with the Periodic Table as the central node and groups like Metals, Non-metals, Noble Gases, etc., as the main branches at the first level. You can also structure the map using rows 1 to 7 of the Periodic Table, for example.
- Alternative Memorization Methods: The Dominic System, Method of Loci, and Memory Palace can be useful alone or in combination with others.
- Starting from Unused Numbers: If you have already memorized the first 600 positions with other information, begin with 601 for Hydrogen, then 602 for Helium, and so on.
- Using Two Matrices: Create a matrix of 18 columns by 7 rows for the elements (excluding Lanthanides and Actinides) and a matrix of 15 columns by 2 rows for Lanthanides and Actinides, using colors to differentiate the groups. The columns can be numbers, and the rows can be letters or numbers, as shown in the table above (click to enlarge).
- Colour-Coded Periodic Table: Use colours to help memorize the elements of the Periodic Table.